Lukas Sodergren and Kaitlyn Bellotti
Synthetic Strategies toward a Cyclic Ethynlene Terphenylene Oligomer as a Carbon Nanotube Precursor
Abstract:
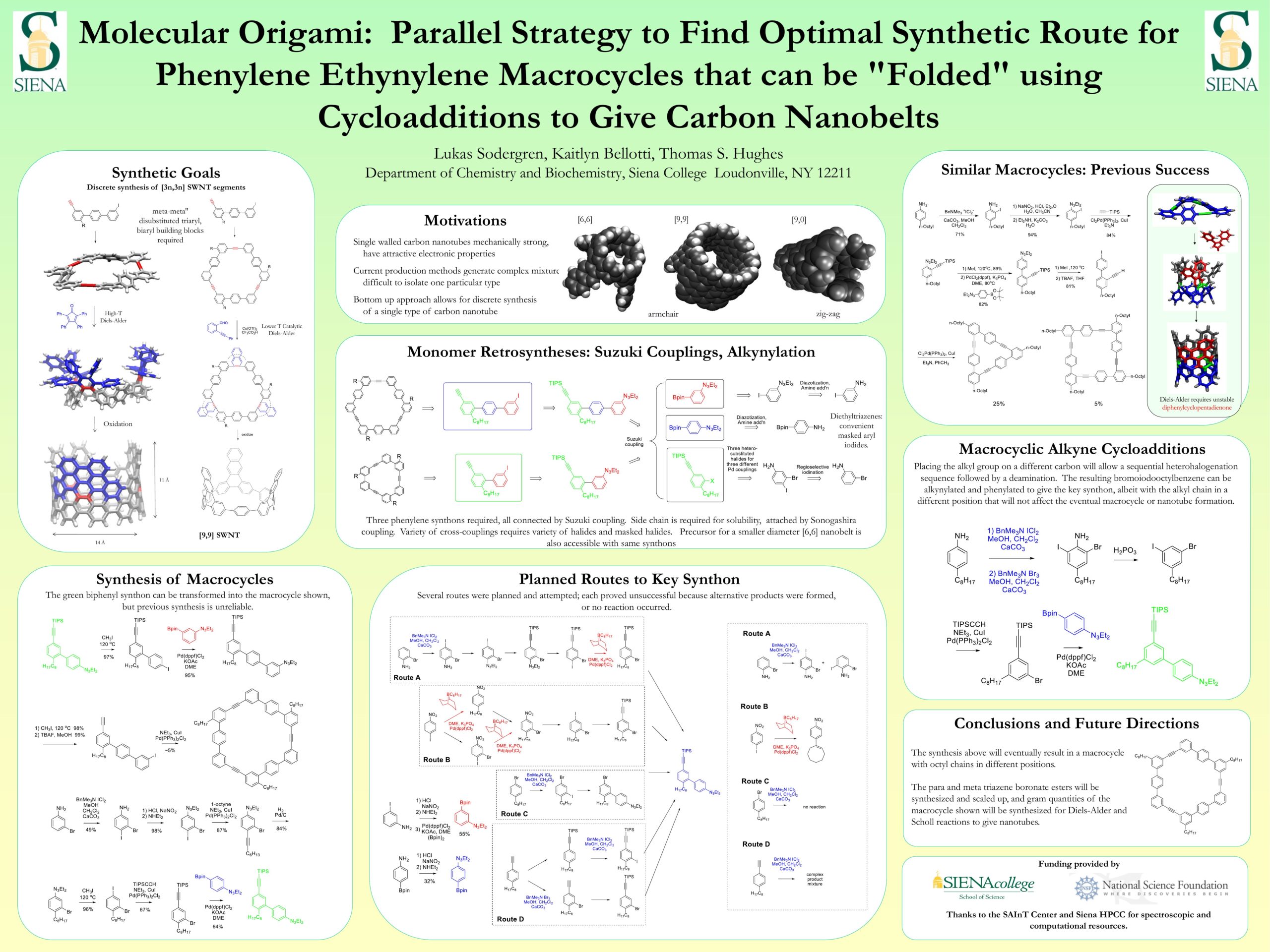
The Hughes Research Group has identified cyclic ethynylene phenylene oligomers as precursors of structurally well-defined carbon nanotubes. This requires the synthesis of an arene that bears an alkyl chain to make the final product soluble, and two halide-equivalent moieties that could be deployed at different times to allow C-C bond formation between the ethynylene and phenylene structures. Several synthetic pathways have been developed and attempted, but they have all suffered from purification challenges and/or inconsistent yields and product selectivities. To surmount the previous challenges, the position of the alkyl group was changed and a sequence of halogenations of 4-alkylaniline followed by deamination was developed and the results of the these halogenations as well as the auxiliary synthons is reported.
Title
Synthetic Strategies toward a Cyclic Ethynlene Terphenylene Oligomer as a Carbon Nanotube Precursor
Faculty Advisor
Dr. Thomas Hughes
Course
Summer Research
Presentation Type
Location
Table 23

